
Regulation of Presynaptic Function & Novel Tools to Study It
The link between electrical activity and chemical neurotransmission is provided by voltage-gated calcium (Ca2+) channels. Presynaptic spikes drive these channels to open and thus allow Ca2+ ions to rush into the synaptic bouton, causing a chemical signal that acts very locally to trigger vesicle fusion. Investigators in the Tsien Lab have made past contributions to the study of presynaptic Ca2+ channels and their regulation by neuromodulators, and adhere to the idea that under most circumstances, transmission requires the opening of multiple channels. Our current interests include questions about how the channels are anchored in appropriate abundance and proximity to the sites of vesicle fusion, and how variations in Ca2+ channel activity regulate synaptic strength.
To aid this understanding, we have developed new optical probes to monitor synaptic vesicle turnover, and by extension, to study synapses and circuits. By creating sypHTomato, a probe attuned to reporting presynaptic activity with red light, Yulong Li and Richard Tsien have opened up the possibility of simultaneously interrogating another probe that emits green light. This second probe could detect another synaptic input, or be responsive to changes in Ca2+ concentration. We show proof-of-principle that dual color recordings, matched with dual color excitation using color variants of channelrhodopsin, could achieve multiplexed, optogenetic stimulation and recording.
Related Publications
Yulong L and Tsien RW. pHTomato, a red, genetically encoded indicator that enables multiplex interrogation of synaptic activity. Nature Neuroscience. 2012. DOI.
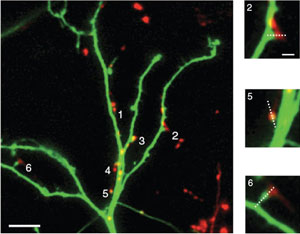
We generated a bright red, pH-sensitive fluorescent protein that can be used to monitor neuronal activity in parallel with green probes like GFP. SypHTomato, made by fusing pHTomato to synaptophysin, can report activity-dependent exocytosis as efficiently as green reporters. Expressing sypHTomato and GCaMP3 in separate cells enables simultaneous determination of presynaptic vesicular turnover and postsynaptic responses in a connected pair of neurons.
Cao Y and Tsien RW. Different relationship of N- and P/Q-type Ca2+ channels to channel-interacting slots in controlling neurotransmission at cultured hippocampal synapses. Journal of Neuroscience. 2010. DOI.
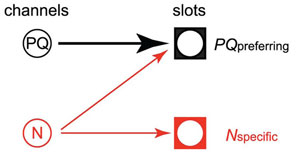
Synaptic transmission at CNS synapses can be mediated by joint actions of multiple channel subtypes. By recording from cultured hippocampal cells overexpressing the Cav2.2 subunit, we dissect the contributions of N-, P/Q-, and R-type channels in excitatory synaptic transmission. We found evidence that indicates two different kinds of presynaptic slots: N(specific) and PQ(preferring).