
Regulation of Germline Proliferation & Differentiation by the Environment: Insulin, TOR & TGFß
Research in the E. Jane Albert Hubbard Lab aims to understand how the insulin, TOR, and TGFß signaling pathways regulate germline proliferation and differentiation.
Diet and Expansion of the Germline Stem/Progenitor Cell Pool
Using largely unbiased genetic approaches, we found that components of the conserved insulin, TOR, and TGFß signaling pathways are important for the expansion of the larval germline stem/progenitor pool that occurs under favorable environmental conditions (Michaelson et al., 2010; Korta et al., 2012; Dalfó et al., 2012; Pekar et al., 2017; Roy et al., 2018).
The larval germline progenitor pool is particularly sensitive to environmental conditions. A dramatic example of this is the newly hatched L1 larva. Normally, if the worms hatch in the absence of food, the worms do not develop and germ cells do not divide. As discovered by Fukuyama et al. (2006), the activity of DAF-18, the sole worm ortholog of the potent human tumor suppressor PTEN, prevents germ cells from proliferating inappropriately in the absence of food. We found that DAF-18 also prevents inappropriate accumulation of transcripts associated with germline genes, and that the regulation of this transcriptional readout differs from that of cell cycle (Fry et al., 2020 BioRxiv).
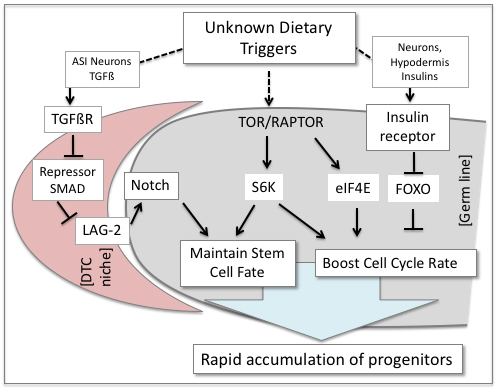
TOR and its downstream effector S6K mediate the effects of food abundance and protein intake on the later larval progenitor pool. We found that the insulin receptor pathway and TOR signaling via S6K are required within the germ line to promote larval cell cycle progression. However, these two pathways do not act in a linear fashion, and their effects on the cell cycle are dissimilar. S6K additionally promotes the undifferentiated germ cell fate: loss of S6K potently enhances phenotypes associated with reduced Notch signaling and suppresses phenotypes associated with elevated Notch signaling (Korta et al., 2012). We recently identified genes that act in concert with S6K to promote the undifferentiated fate (Roy et al., 2018).
We also identified the TGFß pathway as a player in the expansion of the germline progenitor pool through a “neuron-to-niche” signaling axis. TGFß produced in ASI neurons mediates the effects of food and olfactory (pheromone) cues on the germ line. Its action requires a receptor on the distal tip cell (DTC), the germline stem cell niche, not in the germ line itself, to promote expansion of the larval germline progenitor pool (Dalfó et al., 2012). In the DTC, TGFß signaling interferes with the activity of a transcriptional repressor that binds the promoter of a gene that encodes a ligand for the Notch receptor. Therefore, in good conditions, TGFß activity is high, expression of the Notch ligand is high, and the stem cell pool expands (Pekar et al., 2017).
We do not yet know the precise components of the C. elegans diet that act via these pathways.
One way we are currently exploring the interaction between diet—which also refers to the general microbial environment, with all of its potential to communicate both sensory information and nutrients to worms—and stem cells is a two-organism genetic approach that takes advantage of mutant E. coli (the worm’s “lab chow”). Our ongoing collaboration with the Belasco Lab investigates how the genetics of bacteria affects nematode fertility, integrating bacterial genetics, C. elegans reproduction, the microbiome, and nematode parasites.
How Did We Discover That These Pathways Are Important?
We screened for genetic perturbations that interfere with the expansion of the germline progenitor pool during larval development, but that do not interfere with germ cell proliferation itself (Michaelson et al., 2010; Dalfó et al., 2020). The strategy took advantage of our previous work on mutants that exhibit a characteristic proximal germline tumor (Pepper et al., 2003(a,b); Killian and Hubbard, 2004, 2005; McGovern et al., 2009).
In short, we determined that inadequate accumulation of germ cells in larval stages impairs the extent of DTC migration. This occurs since proliferating germ cells contribute to gonadal outgrowth. This outgrowth together with intrinsic DTC migration programs dictate the extent of DTC migration. When early DTC migration is impaired or delayed, this has the secondary consequence of prolonging Notch signaling in proximal germ cells and delaying their differentiation relative to the developmental stage of the rest of the worm. We further discovered that as a result of this delay in germline development, certain newly-born cells in the proximal gonad come into contact with undifferentiated germ cells when normally they would only contact differentiated germ cells.
We then discovered that these proximal somatic cells can act as a “latent niche”: they produce Notch ligands that induce the neighboring proximal germ cells to remain undifferentiated and proliferate. Thus, counterintuitively, interfering with timely and robust expansion of the germline progenitor pool in larval stages can cause tumor formation later in development. The general concept of the latent niche that we uncovered in C. elegans could conceivably underlie certain aspects of human tumorigenesis or metastasis.
This developmental scenario provided a convenient genetic strategy to look for genes that promote expansion of the germline progenitor pool. Since the loss of genes that interfere with robust expansion of the larval progenitor pool can enhance tumor formation in later stages in a genetically sensitized background, we identified such genes by screening for enhancement of tumors and then looking back at earlier stages to examine the effect on the larval progenitor pool. Importantly, this strategy uses germ cell proliferation in the tumor as a read-out, and can therefore identify developmentally important regulators of progenitor pool expansion as opposed to housekeeping genes required for cell proliferation per se (see Hubbard and Schedl 2019 for review).
Relevant Publications
Tolkin T … Greenstein D. Innexin function dictates the spatial relationship between distal somatic cells in the Caenorhabditis elegans gonad without impacting the germline stem cell pool. eLife. 2022. DOI.
Dalfó D … Hubbard EJA. A Genome-wide RNAi screen for enhancers of a germline tumor phenotype caused by elevated GLP-1/Notch signaling in Caenorhabditis elegans. G3 (Bethesda). 2020. DOI.
Hubbard EJA and Schedl T. Biology of the Caenorhabditis elegans germline stem cell system. Genetics. 2019. DOI.
Roy D … Hubbard EJA. Functional interactions between rsks-1/S6K, glp-1/Notch, and regulators of Caenorhabditis elegans fertility and germline stem cell maintenance. G3 (Bethesda). 2018. DOI.
McGovern M … Hubbard EJA. The DSL ligand APX-1 is required for normal ovulation in C. elegans. Dev Biol. 2018. DOI.
Pekar O … Hubbard EJA. Linking the environment, DAF-7/TGFβ signaling and LAG-2/DSL ligand expression in the germline stem cell niche. Development. 2017. DOI.
Roy D … Hubbard EJ. Functional interactions between rsks-1/S6K, glp-1/Notch, and Regulators of Caenorhabditis elegans fertility and germline stem cell maintenance. G3 (Bethesda). 2018. DOI.
Hubbard EJ and Schedl T. Biology of the Caenorhabditis elegans germline stem cell system. Genetics. 2019. DOI.
Roy D … Hubbard EJ. Cell cycle features of C. elegans germline stem/progenitor cells vary temporally and spatially. Dev Biol. 2016. DOI.
Hubbard EJ … Dalfó D. Physiological control of germline development. Adv Exp Med Biol. 2013. DOI.
Hubbard EJ … Dalfó D. Sensory regulation of the C. elegans germline through TGF-β-dependent signaling in the niche. Curr Biol. 2012. DOI.
Korta DZ … Hubbard EJ. S6K links cell fate, cell cycle and nutrient response in C. elegans germline stem/progenitor cells. Development. 2012. DOI.
Hubbard EJ. Insulin and germline proliferation in Caenorhabditis elegans. Vitam Horm. 2011. DOI.
Korta DZ and Hubbard EJ. Soma-germline interactions that influence germline proliferation in Caenorhabditis elegans. Dev Dyn. 2010. DOI.
Michaelson D … Hubbard EJ. Insulin signaling promotes germline proliferation in C. elegans. Development. 2010. DOI.
McGovern M … Hubbard EJ. A "latent niche" mechanism for tumor initiation. Proc Natl Acad Sci USA. 2009. DOI.
Killian DJ and Hubbard EJ. Caenorhabditis elegans germline patterning requires coordinated development of the somatic gonadal sheath and the germ line. Dev Biol. 2005. DOI.
Killian DJ and Hubbard EJ. C. elegans pro-1 activity is required for soma/germline interactions that influence proliferation and differentiation in the germ line. Development. 2004. DOI.
Pepper AS … Hubbard EJ. The establishment of Caenorhabditis elegans germline pattern is controlled by overlapping proximal and distal somatic gonad signals. Dev Biol. 2003. DOI.
Pepper AS … Hubbard EJ. Genetic analysis of Caenorhabditis elegans glp-1 mutants suggests receptor interaction or competition. Genetics. 2003. DOI.