
About the Center for the Investigation of Environmental Hazards
At NYU Langone’s Center for the Investigation of Environmental Hazards, we build on our expanded capacity in assessment of organic contaminant exposures, metabolomics and bioinformatics, and clinical and environmental health human studies in three areas:
- integration of organic contaminant exposure assessment into human and laboratory studies
- execution of multiomic studies incorporating a fuller exposome perspective
- broader examination of the effects of environmental exposures beyond the perinatal period
Translational Research Vision and Track Record
With our broad base of investigators, who span disciplines including toxicology, exposure science, epigenetics, community outreach, health economics, and clinical medicine, we have built a strong track record of moving across the five rings of the National Institute of Environmental Health Sciences (NIEHS) translational research framework.
Translational Research Examples
The following examples illustrate synergy across our NIEHS Center, and highlight our ability to evolve research in the context of emerging environmental health issues while maximizing the impact of our work in support of the NIEHS strategic plan.
Endocrine-Disrupting Chemicals
Leonardo Trasande, MD, MPP, director of the center, has performed studies leveraging the Generation R First (R01ES022972) and Next (R01ES029779) to examine prenatal and preconception effects, respectively, of phthalate and bisphenol exposures on fetal and postnatal growth. With support of the Environmental Influences on Child Health Outcomes (ECHO) Program, Dr. Trasande is examining these exposures as well as organophosphate pesticides and polycyclic aromatic hydrocarbons in pregnancy (UH30223305) on fetal and postnatal growth in the Children’s Health and Environment Study and the Infant Development and Environment Study (NIEHS Fundamental Questions ring, population observation).
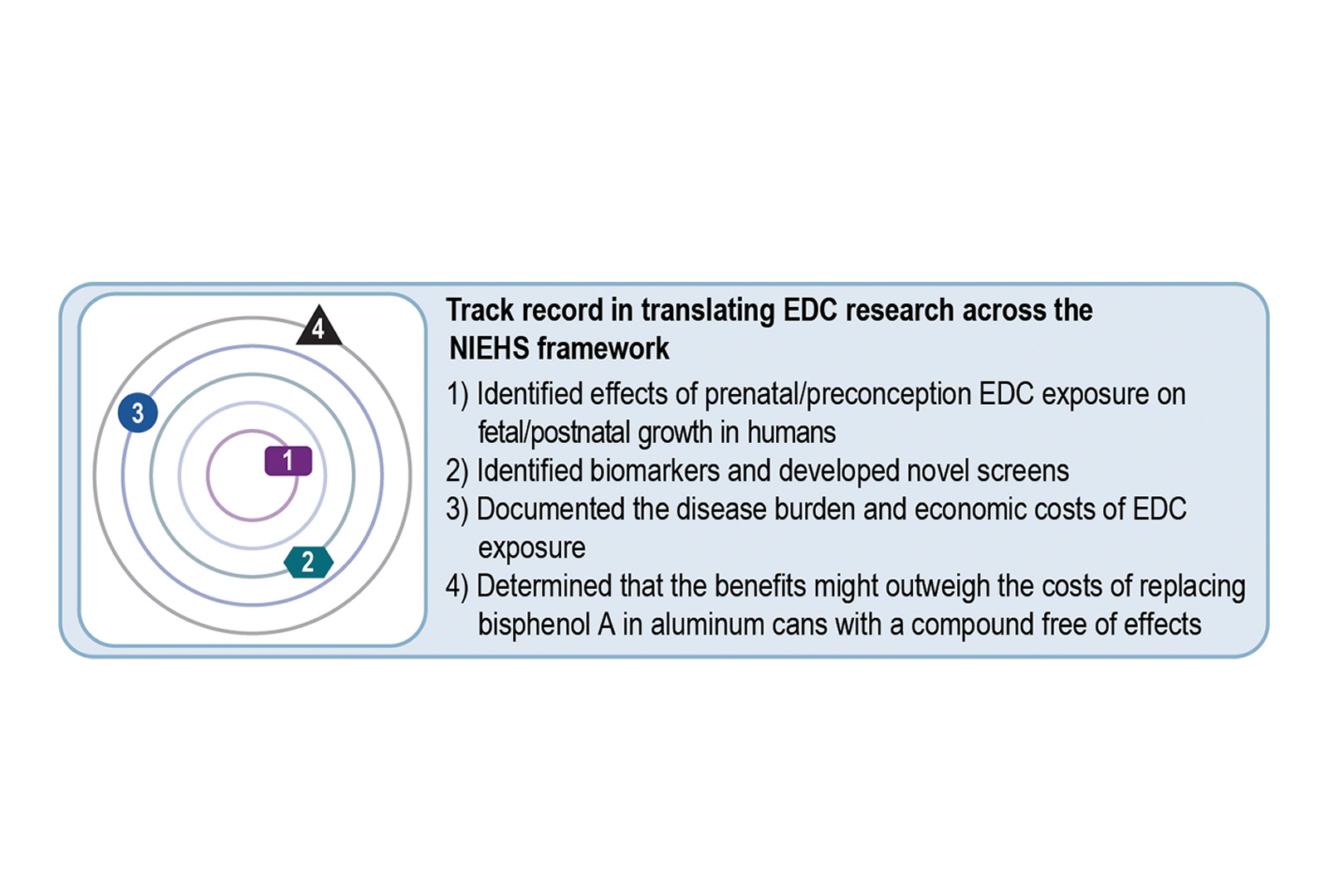
Dr. Trasande has documented substantial disease burden and economic costs due to phthalates, bisphenols, and other endocrine-disrupting chemicals that have informed policy discussions in the United States and Europe (NIEHS Practice ring). He has documented how the economic benefits of replacing bisphenol A (BPA) in aluminum cans with a replacement free of effects may outweigh the costs of such a replacement (NIEHS Impact ring).
Air Pollution
Despite significant progress since the passage of the Clean Air Act in 1970, New Yorkers still breathe polluted air that adversely affects their health. Most people know smog from cars and buses is a major culprit, but a significant amount of dangerous air pollution also comes from the buildings in which we live and work. In the basements of thousands of large residential, commercial, and institutional buildings in Manhattan, Brooklyn, Queens, and the Bronx, boilers burn a dirty fuel to heat their units. This type of oil—referred to as “residual fuel oil” because it is essentially the leftovers from the petroleum distillation process—releases soot and toxic chemicals into the air. Over time, these can lead to cardiovascular disease, asthma, and even premature death.
Studies by Lung-Chi Chen, PhD, and Terry Gordon, PhD, found that numerous trace elements in heating oil are associated with the reactive oxygen species (ROS) response in vascular endothelial cells (NIEHS Fundamental Questions ring). Kevin R. Cromar, PhD, and George D. Thurston, ScD, combined these and other population studies to model health and economic benefits of mitigation (NIEHS Application and Synthesis, and Implementation and Adjustment rings). It was found that converting residential, commercial, and institutional boilers from residual oil to natural gas would decrease the soot New Yorkers breathe, avoiding hundreds of deaths and other adverse health impacts, and saving billions of dollars in associated health costs.
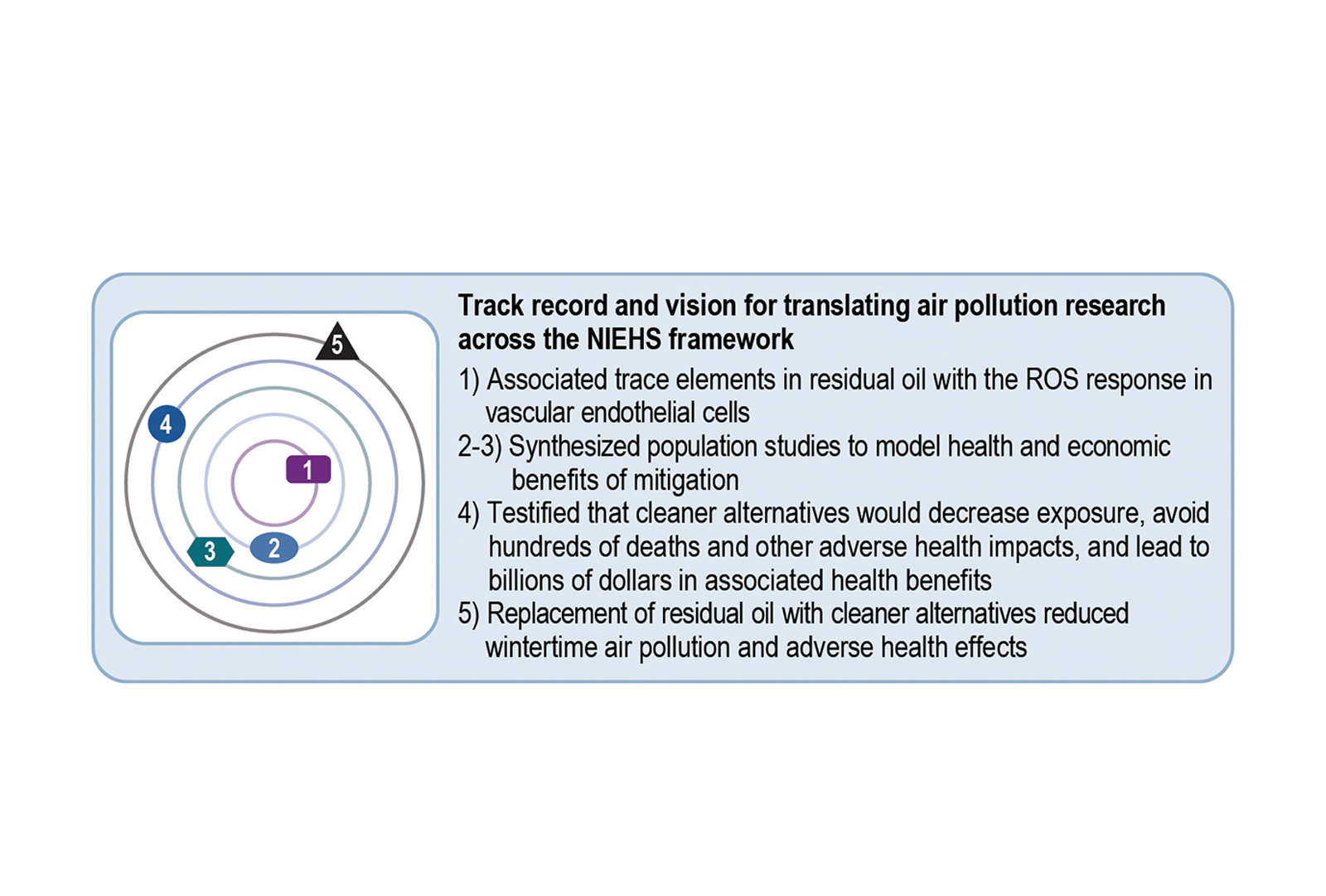
Dr. Cromar worked closely with the New York City Department of Health and Mental Hygiene and testified on this matter before the New York City Council, leading to new policy implementation in New York City (NIEHS Practice ring). These findings contributed to a subsequent ban in New York City on the use of residual oil in favor of cleaner oil and natural gas. Then-mayor Michael Bloomberg allocated more than $100 million to help buildings convert to clean heating fuels, leading to a substantial reduction in winter air pollution and adverse health effects in New York City (NIEHS Impact ring).